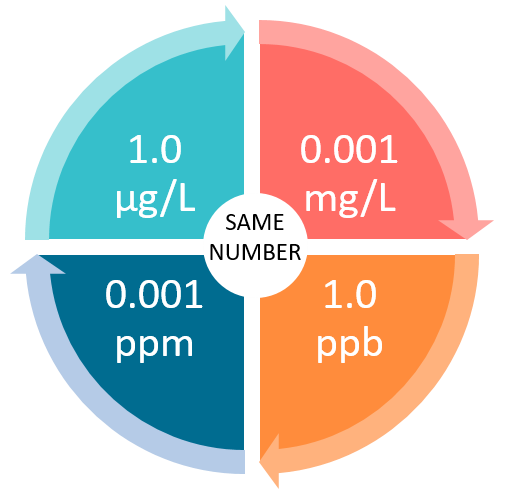
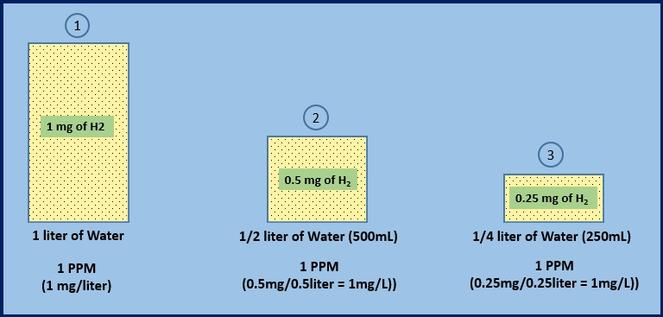
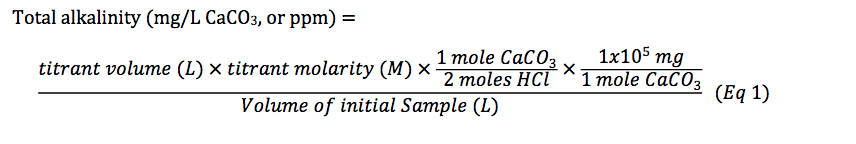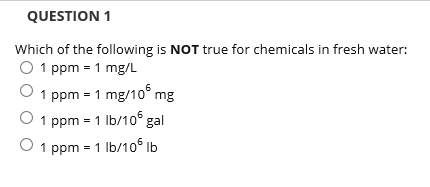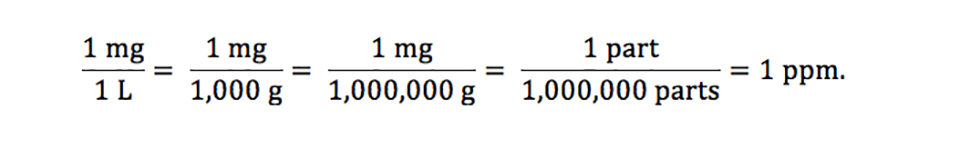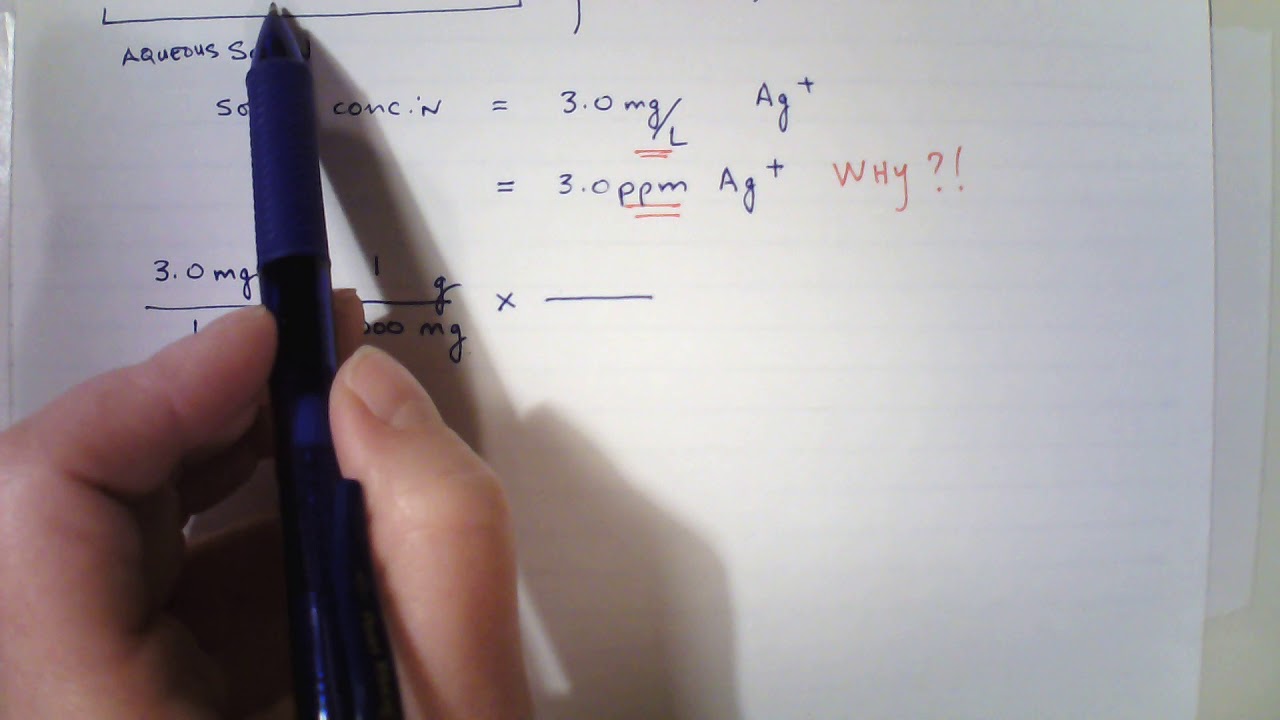SOLVED: A 148-mg sample of Na2CO3 is dissolved in H2O to give 1.70L of solution. What is the concentration of Na+ in parts per million (ppm)? Note: "ppm" are 106 ×[(mass of

SOLVED: How many grams of mgco3 dissolved per litre gives 84 ppm hardness? A. 70.56 mg/l b. 48.23 mg/l c. 81.49 mg/l d. 66.12 mg/l